Recently, my family made a trip to Detroit with 9 kids in tow. We only had two days to spend. Here are some of the things we did:
We were on a tight budget for time and had lots of squirmy little ones, so we didn’t do any fancy dining, even though Detroit has a vibrant & competitive food scene. We ordered delivery of Detroit style pizza and took in the food trucks at Eastern Market. In addition to good food and local items (need a t-shirt?), Eastern Market is known for its murals . Art in Detroit is a combination of quirky and hopeful. Seriously, click that link.
We decided to take the Coney Dog Challenge. Despite the name, Detroit, not Coney Island, invented the coney dog (chili sauce, onions, and mustard). Two famous coney dog spots are right next to each other downtown on Lafayette Boulevard. After trying both hot dogs, what was our verdict? It was mixed. Some liked American because the dogs have a great snap to them and a delightful casing, while others found the sauce of Lafayette more savory. American has a little more room to sit and eat.

We stopped by Sanders, in the wealthy suburb of Grosse Pointe, for ice cream, Vernors, and chocolate.





- Campus Martius is Detroit’s downtown hub of activity and is the aptly marked origion of the city. The summer fun was a beach with sand and toys, plus an outdoor bar which a couple harried parents appreciated. We wandered to the riverfront, past more eye caching art work. It was a beautiful moment in The D.






Shown above are the Civil War Memorial–Michigan Soldiers’ and Sailors’ Monument (1872)
KAWS statue, Waiting (2018)
Spirit of Detroit (1958)
Monument to Boxer and Civil Rights Icon Joe Lewis (1986)
- Belle Isle is the oldest aquarium and botanical garden in the United States. It holds the world’s most complete collection of gar fish. It was a quick and enjoyable visit. The kids not only marveled at the aquatic life, they lingered over the plants, everything from cactuses to Dumb Cane to Banana Trees.





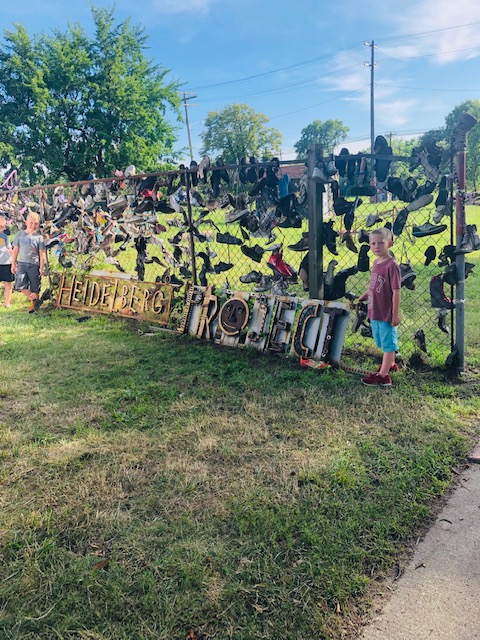
- The Heidelberg Project (shown above) is a found object art installation that has become increasing popular. And for good reason. It’s a dynamic area and the kids were inspired to give each peice their own interpretation.
- The Henry Ford Museum in Dearborne is the opposite of the Belle Isle attractions. It contains everything made by humans to help them travel plus an extenive array of historical furnature and historical items, including the Rosa Parks bus. We need to go back and take in the rest of this sprawing attraction.


Detroit is a center for art and social change. I hope it continues to thrive and keep its hopeful, quirky nature. The kids loved it, had discussions about art and Civil Rights, and one who is a city girl kept remarking about the tell buildings, even though she sees tall buildings in her city. Pure Detroit gives some great tours if you want to explore some of the impressive Detroit buildings.


















































